Targeted Therapy
Targeted therapy is directed against specific molecules on certain cells. The therapy, based on e.g. antibodies can function in a number of ways. The specific antibody can for example block the growth of cancer cells by interfering with specific signaling molecules, or carry a radionuclide or a toxin that can kill the tumor cell. This is in contrast to conventional therapy, such as chemotherapy, that is none specific and affects all dividing cells. Consequently, targeted therapies can in many cases be more effective for the patient, be patient specific based on pre-therapy imaging/dose planning and is often associated with fewer side effects.
Within the MAD for Cancer Program, there are more than 20 years experiences of therapeutic antibodies based on phage library design. These technologies can be used in targeted therapy and have during the past decades revolutionized the ability to develop highly efficient therapeutic antibodies without engaging an in vivo immune system.
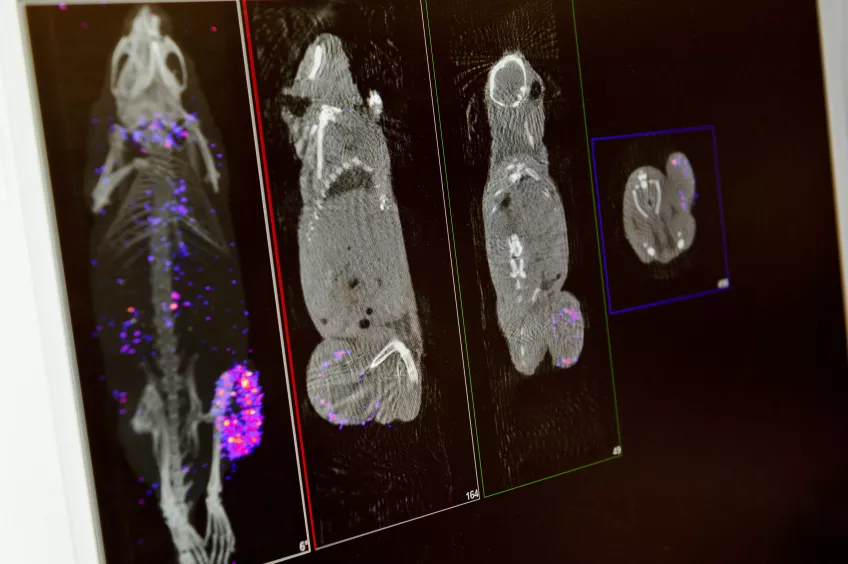
Advanced imaging systems used for improved monitoring of treatment and to further develop strategies for targeted therapy is a focus at the Lund University Bioimaging Center (LBIC), established in 2008. LBIC has pioneered the research on radioimmunotherapy and the center provides access to experimental SPECT/CT and PET/CT equipment. From mid-2015, LBIC hosts a unique ultra-high-field 7T MRI scanner for human research giving the center a National status and an internationally leading position in medical imaging.
Using their in-house developed or other novel strategies for pre-clinical radioimmunotherapy or radionuclide therapy, LBIC now aim to employ microarrays to investigate cell response and transcriptomic variations in human tumor xenograft models. This would give a much broader understanding of organ radiosensitivity, tumor response and radiobiology research, which translates into patient value.